Refitting Success: Reducing Suction and Maximizing Oxygen Permeability
By Karen Carrasquillo, OD, PhD, FAAO, FSLS, FBCLA
Case Summary
Case of a 60-year-old woman with a history of keratoconus and dry eye syndrome in both eyes, history of corneal scraping in the left eye 25 years prior, history of post hydrops left eye, and questionable potential history of amblyopia left eye. She had a history of successful PROSE treatment since 2010 (11 years of PROSE device wear) but underwent penetrating keratoplasty (PKP) in the left eye in December 2019; secondary to diffuse opacification (Figure 1) and limited best corrected visual acuity.
Prior to PKP in the left eye, she had been wearing 18.5mm, quadrant-specific PROSE devices in both eyes, with added front-surface astigmatism in the right eye and with resultant best corrected visual acuity (BCVA) of 20/30, PH 20/25-2 and 20/100, PH 20/60–, right eye and left eye respectively. With prescribed polycarbonate spectacles to correct residual astigmatism in the left eye and to serve as protection (especially for the better-seeing eye), BCVA in the left eye could be improved to 20/70. The patient wore the spectacles as needed, depending on daily activities and especially during night driving.
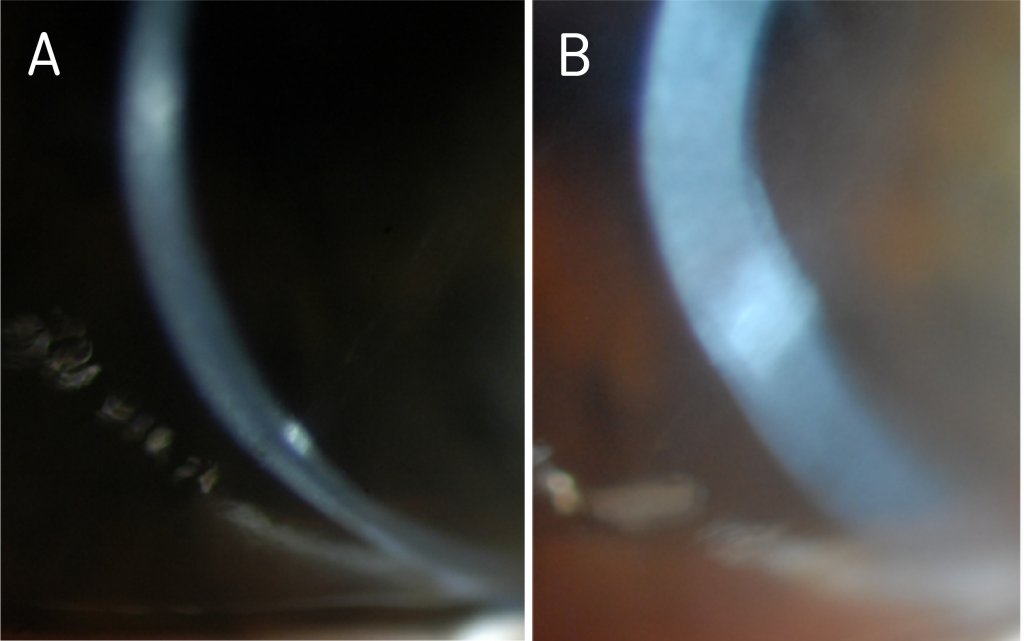
Figure 1. Diffuse haze and opacification in the left eye, as seen on optic section (A) and wide beam (B), prior to PKP.
After corneal transplantation and having been cleared to resume scleral device wear in the left eye, she was referred to our clinic for refitting.
Evaluation After Penetrating Keratoplasty
Initial evaluation after PKP OS (Topography shown in Figure 2A), showed a clear and compact graft, with interrupted sutures (Figure 2B). There was no evidence of subepithelial infiltrates, microcystic edema, no keratic precipitates, or stromal diffusion after corneal staining. The patient was on Prednisolone acetate 1% gtts QD and as sutures were removed, she was prescribed Moxifloxacin 0.5% TID in the left eye. Evaluation was also remarkable for cataracts in both eyes, not visually significant at the moment. Best corrected vision during this visit in the right eye was 20/40, PH 20/30-2 and uncorrected visual acuity in the left eye was CF at 3’, PH 20/200.
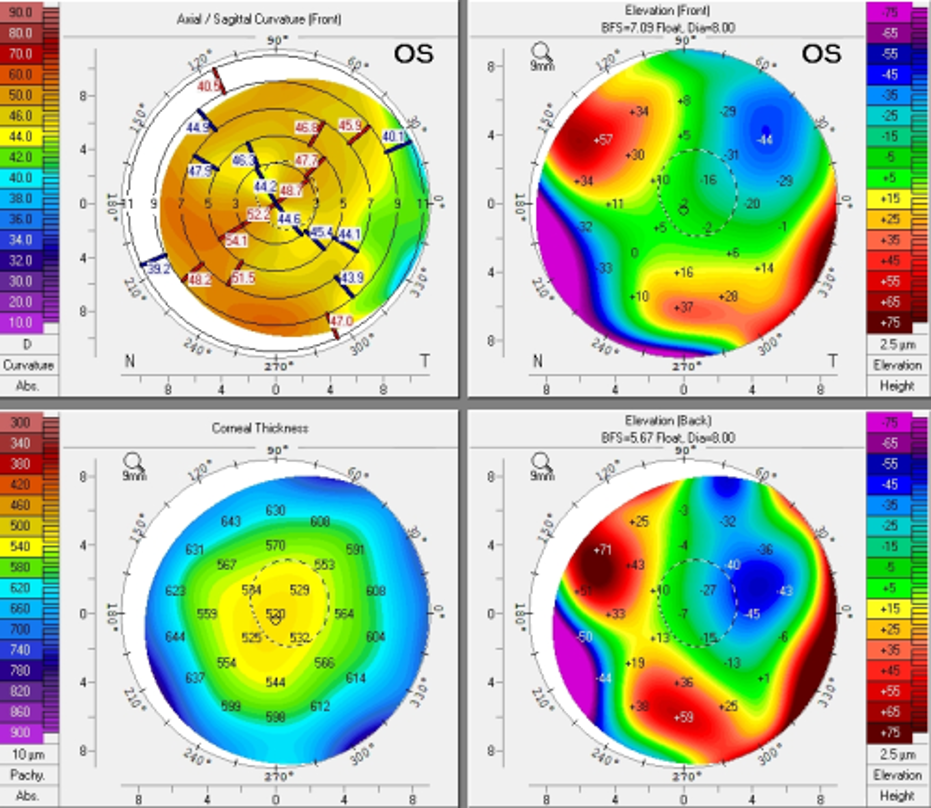
Figure 2A. Pentacam images s/p PKP in the left eye.
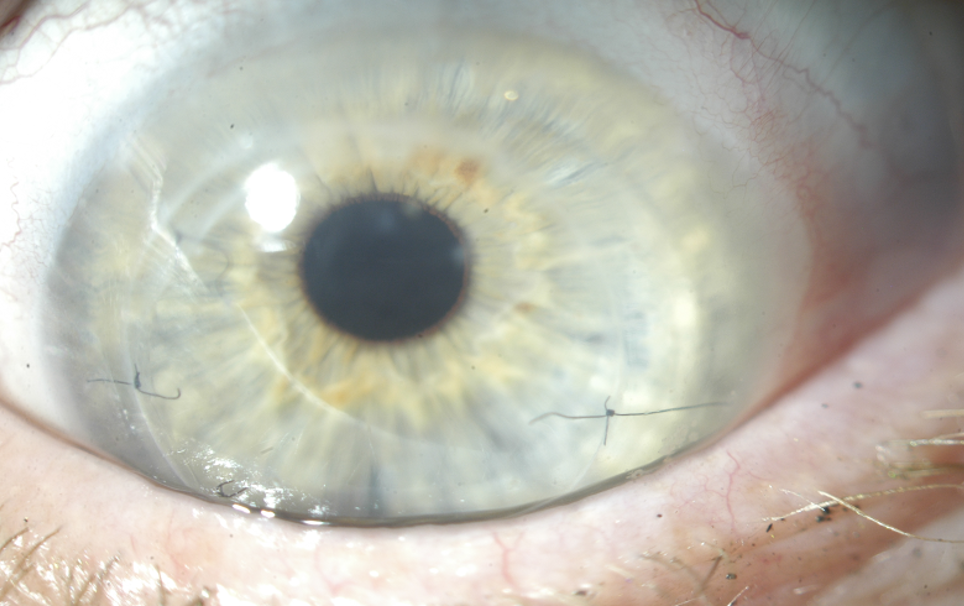
Figure 2B. Presentation of left cornea, status post corneal graft during baseline evaluation for refitting.
Refitting
We started a new fit, deciding to keep the patient in the habitual diameter of 18.5mm. Because the cornea was now grafted, we opted to design the lens using Optimum Infinite material (180 Dk) so that together with a center thickness of 0.25mm, oxygen permeability was maximized. Best documented corrected visual acuity in the left eye was 20/30-2 with the new 18.5mm device. While the patient was elated about the newfound visual potential, she reported sensation of pressure during device wear in the left eye – despite no objective evidence of significant compression at the haptics (Figure 3).
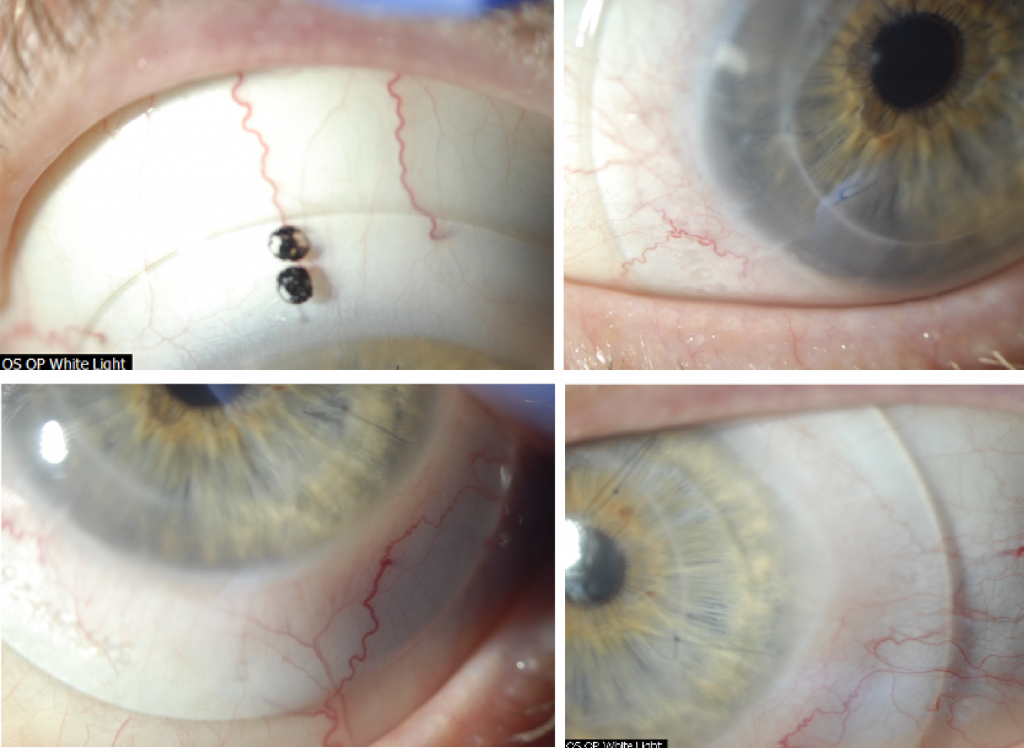
Figure 3. Initial fitting endpoint with an 18.5mm PROSE device after PKP OS.
An attempt was made to flatten the haptics, modify the area along the mid-haptic portion to minimize mid-haptic compression and obtain better alignment. Despite these efforts, the patient continued to report pressure sensation with device wear and difficulty with device removal at the end of the day. These symptoms are traditionally associated with lens compression and suction with increased wear time. After reviewing the findings and the thought process with the patient, the decision was made to increase the lens diameter to 19.5mm to provide a larger surface area on which the device could rest. Figure 4 shows the new fitting endpoint with the 19.5mm, 8-meridian quadrant-specific device. One can observe good haptic alignment and the eye is white and quiet, with a nice and even distribution of lens clearance over the grafted cornea.
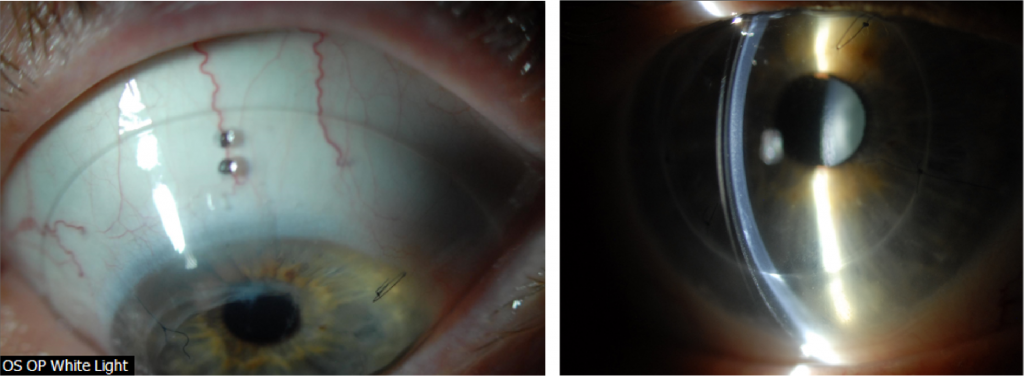
Figure 4. Fitting endpoint with an 8-meridian, quadrant-specific 19.5mm PROSE device after PKP OS.
Comparison OCTs show the increased landing surface area when going from an 18.5 (Figure 5) to a 19.5mm device (Figure 6). This attempt to maximize weight distribution over a larger surface area has proven efficacious in our clinical practice when dealing with compression symptoms and device intolerance. While maintaining the same sagittal height, we additionally flattened the base curve to accommodate the more oblate nature of the cornea after transplant as shown in Figure 7.
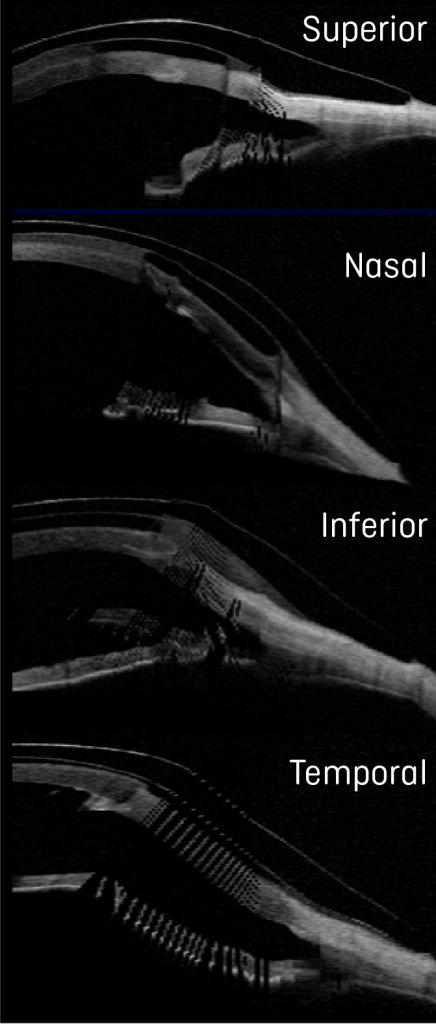
Figure 5. Ocular Coherence Tomography (OCT) of an 18.5mm device in the different orthogonal meridians of the left eye.
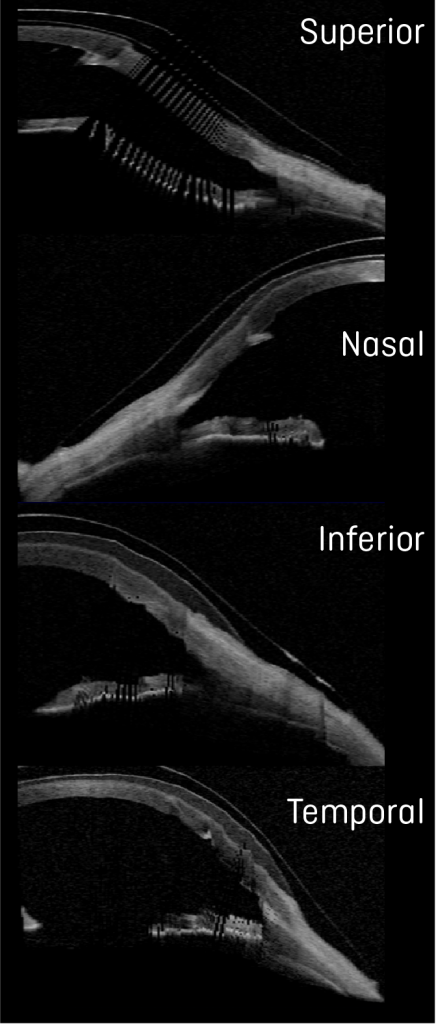
Figure 6. Ocular Coherence Tomography (OCT) of a 19.5mm device in the different orthogonal meridians of the left eye.
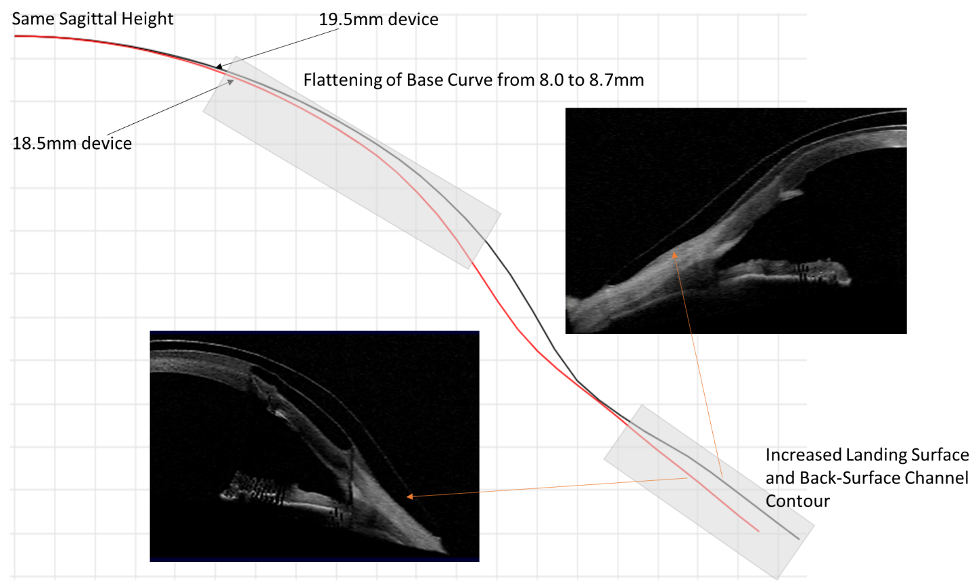
Figure 7. Graphical images of 18.5 and 19.5mm, juxtaposed to show the changes to the lens design based on parameter changes using spline curve technology and as observed by OCT. The nasal meridians corresponding to the flattest meridian in the eye are shown for comparison.
Interestingly, while we were optimizing the fit and after reporting improvement in comfort with increased lens size, the vision in the left eye dropped to the 20/50-20/60 range over the course of a couple of months. During all evaluations, we carefully monitored for signs of microcystic epithelial corneal edema, signs of rejection, etc. and at no point were evidence of these found. Given the history of previous cataracts and the fact the patient was on chronic low dose steroids, we performed an undilated assessment of the human lens in the left eye. Photo documentation revealed the development of a posterior subcapsular cataract (PSC) (Figure 8), which was later confirmed to be visually significant during dilated assessment by her corneal specialist. The patient decided to move forward with cataract extraction with hopes to improve vision.

Figure 8. Evidence of PSC formation, during undilated assessment.
Before cataract surgery, we decided to challenge the current 19.5mm design hoping to obtain a comfortable and physiologically compatible lens design. After the patient wore the lens for one week, she reported that the previous symptoms of pressure and difficulty with lens removal had improved, but they were still present. To further minimize compression and suction symptoms, we added 4 back-surface haptic channels to soften further the landing of the haptic surface from bearing harshly on the conjunctiva and prevent a seal off effect (Figure 9).

Figure 9. A 19.5mm, 8-meridian quadrant-specific PROSE device with the addition of 4 back-surface haptic channels to minimize suction and improve comfort.
Discussion and Final Outcomes
Long-term outcomes and benefits of scleral lenses after penetrating keratoplasty have been reported in the literature – while most patients benefit from improvement in visual acuity, complications with scleral lenses, i.e. microcystic corneal edema can ensue in this patient population.1 Special consideration should be taken when fitting or refitting grafted corneas with scleral lenses to minimize the risk of corneal edema, erosions and ultimate rejection. Strategies to promote fluid-ventilation like avoiding tight-fitting edges, lens seal-off and adding back-surface haptic channels can be especially important.1-5 Clinical practice has also shown that the addition of back-surface channels can have the potential to minimize lens suction over time and lead to improvement in physiological endpoints.5-7
In this case, with an increase in scleral lens landing surface area, customized haptic in an 8-meridian quadrant-specific manner and the addition of 4 back-surface haptic channels, not only are we promoting fluid-ventilation to optimize physiological endpoints, but we have a reported significant improvement in comfort. The choice to manufacture lenses in Optimum Infinite material potentiates the oxygen permeability to the grafted cornea. With the final lens after cataract surgery, the patient was able to achieve 20/25+2 best corrected visual acuity with an average comfortable daily wear of up to 12 hours.
By undertaking strategies to progressively soften the landing over the conjunctival surface to reduce or eliminate lens compression over time and minimize suction we were able to achieve a successful refitting outcome.
1. Severinksi, B. et. al. (2014) Scleral contact lenses for visual rehabilitation after penetrating keratoplasty: long-term outcomes. Cont Lens Ant Eye, 37, 196-202.
2. Rosenthal, P. Croteau, A (2005) Fluid-ventilated, gas-permeable scleral contact lens is an effective option for managing severe ocular surface disease and many corneal disorders that would otherwise require penetrating keratoplasty. Eye Cont Lens, 31(3), 130-134.
3. Severinksy, B et. al. (2010) Current applications and efficacy of scleral contact lenses – a retrospective study. J Optom, 3(3), 158-163.
4. Stason WB, Razavi M, Jacobs DS, et al. (2010) Clinical benefits of the Boston ocular surface prosthesis. Am J Ophthalmol, 149, 54–61.
5. Carrasquillo, KG, Byrnes, S (2018) Corneal edema and scleral lenses. Cont Lens Spect, 33, 34-41.
6. Barnett, M (2018) Mind the suction. Scleral Lens Monthly, Nov 2018.
7. Carrasquillo, KG (2020) Mind the suction II. Scleral Lens Monthly, Jul 2020.
Thank you to Dr Karen G.Carrasquillo for contributing to Global Insight.

Dr. Karen G. Carrasquillo is Vice President of Clinical and Professional Affairs at BostonSight. In addition, she is an adjunct clinical professor at the New England College of Optometry, an adjunct clinical professor at the school of optometry, MCPHS University, Advisory Board member for the Gas Permeable Lens Institute (GPLI), Fellow of the American Academy of Optometry (FAAO), Fellow of the Educational Society of Scleral Lenses (FSLS), and Fellow of the British Contact Lens Association (FBCLA). She is the founder and Program Chair of FitAcademy, an educational retreat for Cornea and Contact Lens Residents, and is also a planning committee member for the International Congress of Scleral Contacts (ICSC) Meeting and Global Ophthalmic Women (GLOW) meeting.
