Dry Eye and Scleral Lenses
By Dr. Elise Kramer, OD, FAAO, FSLS
A 71-year-old man presented to the Miami Contact Lens Institute for a scleral lens refit. He was wearing his scleral lenses but complained of significant lens awareness and foggy, dirty lenses. He reported having worn the lens only four times since they were originally dispensed because of discomfort. A review of his ocular history revealed severe keratoconjunctivitis sicca caused by Sjögren’s syndrome and acquired toxoplasmosis of the left eye. The patient reported that the latter was no longer active but that owing to a recent flare-up, he had retinochoroiditis, leaving him with very poor central vision in that eye. He had no history of glaucoma or trauma and no family history of ocular disease. In addition, he had a history of hypertension, hyperlipidemia, anxiety, depression, and prostate cancer.
He had been taking 20 mg of Plaquenil daily for the last 10 years, prescribed for his Sjögren’s syndrome. For his dry eye symptoms, he was using over-the-counter artificial tear drops, autologous serum eye drops and topical steroids, and had had plugs inserted in all four punctae. Despite these treatments, the patient continued to have severe ocular dryness, for which scleral lenses had been fitted. Visual acuities with his current scleral contact lenses (ScCLs) were 20/60 in the right eye and 20/400 in the left eye. His pupils were equal, round and reactive to direct and consensual illumination, and no afferent pupillary defect was observed in either eye. Confrontational visual fields were full to finger counting in both eyes, and extraocular motility was full in both eyes. The patient had moderate scurf in his eyelashes, which he had been cleaning daily using OcuSoft lid wipes. He had moderate capping of his Meibomian glands, white and quiet conjunctivae, flat irides and clear lenses bilaterally. Slit lamp examination revealed scleral lenses in place in both eyes with sizeable zones of poor wettability, protein and lipid build-up centrally. (Figure 1)
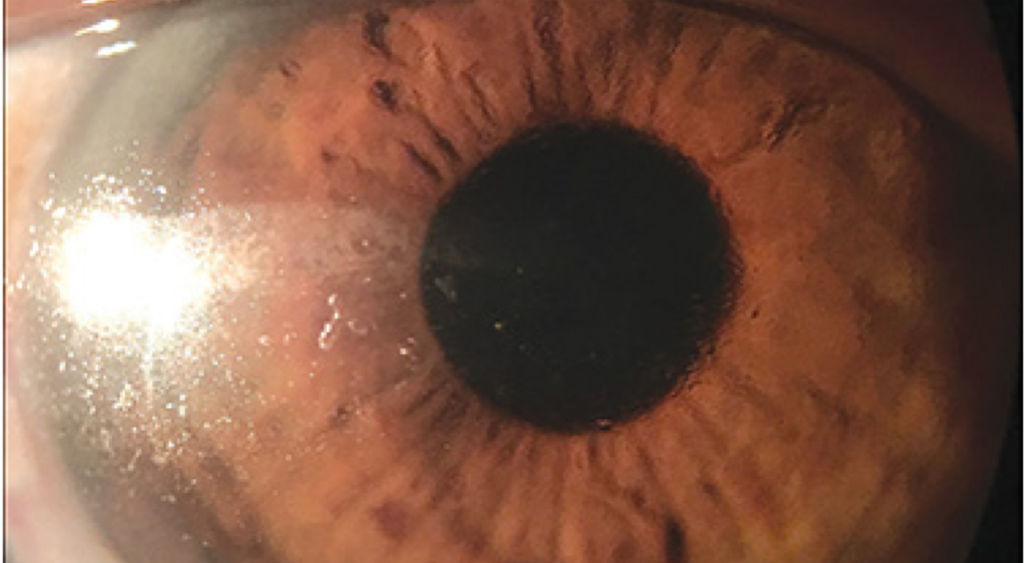
Figure 1. Right eye with scleral lens in place showing significant deposits on the temporal and central portion of the lens.
After the lenses were removed, the ocular surface was examined with a slit lamp. The corneas had mild diffuse staining, as did the conjunctivae. The patient had a low tear meniscus in both eyes. He had flat irides and mild nuclear sclerosis in both eyes. No proptosis or lid abnormalities were observed in either eye. After instilling one drop of 0.5% proparacaine hydrochloride ophthalmic solution at 11:00 AM, intraocular pressures were measured by Goldmann applanation tonometry as 13 mm Hg OU. On dilated fundus examination (after instilling one drop of a solution of 1% tropicamide and 2.5% phenylephrine OU), the cup-to-disc ratios were 0.2 OU. The neuroretinal rims were healthy and pink in both eyes. The macula was clear and flat in the right eye, but the left eye had a very large, white/yellow scar extending from the macula to the peripheral vascular arcades. The vitreous was clear in both eyes, with no signs of active inflammation. The retinal vasculature was of normal course and calibre in both eyes, and no breaks were observed in the retinal periphery.
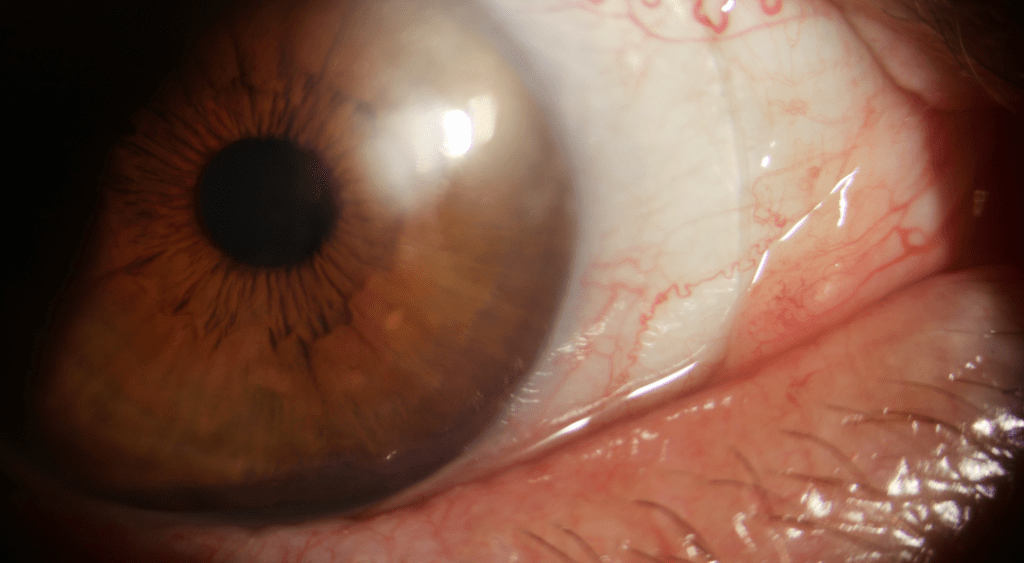
Figure 2. Right eye showing new scleral lens with smooth surface free of deposits
Discussion
Nearly five million Americans over the age of 50 suffer from dry eye. Thirty million, in general, have symptoms of dry eye disease but only 16 million are diagnosed, and one million treated.1 In fact, it is the number one reason that patients visit their eye care practitioners.2 There is huge potential to address this highly prevalent condition. Optimal management can be achieved by basing the treatment on the etiology and severity of the dry eye3 but can be very challenging in severe cases.4 Scleral lenses are now well established in treating dry eye disease, especially in refractory cases where traditional treatments have limited efficacy.4 Current literature recommends the use of contact lenses for Grade 3 dry eye,5 i.e., chronic dry eye that affects comfort and vision.3 They should be considered as a management option when conventional treatments are insufficient and after having tried prescription medications and overnight treatments such as ointments or moisture goggles, and before long term use of steroids, amniotic membrane grafts, surgical punctal occlusion or other procedures such as tarsorrhaphy or salivary gland transplantation.3
Not all lenses are appropriate for severe dry eye, however. In fact, some can do more harm than good. Scleral lenses can be beneficial in these cases by promoting patient comfort and visual function.4
These lenses are custom-made for each patient and are fitted in a way to vault the cornea and maintain a constant reservoir of fluid between the lens and cornea to ensure that the cornea remains hydrated. This fluid reservoir acts as a liquid bandage that protects the ocular surface from the environment and from friction caused by blinking the eyelids. It also compensates for surface irregularities, leading to improved vision. Other types of contact lenses may be limited by the compromised state of the ocular surface. Scleral lens designs can overcome these challenges. They work especially well for vision correction because the fluid layer helps smooth out defects caused by dry eyes, providing a more uniform refractive surface.
The material chosen in the case presented was Optimum Extra (Contamac), which is specifically indicated in dry eye syndrome and provides the comfort of a soft lens with higher optical quality.6 Its DK of 100 allows the lenses to be permeable to oxygen. The coating that was added to the lenses (Tangible Hydra-PEG™) is an advanced contact lens coating technology that encapsulates Contamac Optimum’s gas-permeable material in an ultra-thin layer of PEG-based polymer. The result is called Optimum | Tangible Hydra-PEG™. It creates a highly wettable and lubricated surface, with increased surface water retention due to the polyethylene glycol-based polymer’s high water content (90%) and resistance to protein and lipid deposits, which reduces friction between the eyelid and contact lens. It is therefore particularly suited to contact lens wearers who suffer discomfort due to a number of physiological conditions, such as dry eye.6
In summary, by upgrading the material of the patient’s lenses and adding the Tangible Hydra-PEG™ coating, we were able to improve his comfort and increase his wear time. He was able to wear the lenses comfortably for 12-16 hours per day and reported substantial improvement in his dry eye symptoms. With the increasing recent interest of clinicians and manufacturers, scleral lenses have become “mainstream” in contact lens practice. Dry eye diagnosis and management have also had an increasing interest amongst practitioners. Eye care practitioners should continuously update their expertise in the area of scleral lens design, which can be an excellent management strategy for patients with dry eye and many other ocular problems.
1. The epidemiology of dry eye disease: report of the Epidemiology Subcommittee of the International Dry Eye WorkShop (2007). Ocul Surf. 2007 Apr;5(2):93-107.
2. Gayton JL. Etiology, prevalence, and treatment of dry eye disease. Clin Ophthalmol. 2009;3:405-412.
3. DeNaeyer G. Scleral Lenses: An Overlooked Fix for Dry Eye? Review of Cornea and Contact Lenses 2013, April.
4. Bavinger JC, DeLoss K, Mian SI. Scleral lens use in dry eye syndrome. Curr Opin Ophthalmol. 2015 Jul;26(4):319-24.
5. Jones, L. Downie, LE. Korb, D, et al. TFOS DEWS II Management and Therapy Report. The Ocular Surface. 2017 Jul; 15(3):575-628. Doi:10:1016/j.tos.2017.05.006.Epub
6. Contamac, www.contamac.com/product/optimum.
Thank you to Dr. Elise Kramer, O.D. for contributing to Global Insight.

Dr. Elise Kramer OD, FAAO, FSLS
Optometrist and Contact Lens Specialist
Residency Coordinator, Scleral Lens Education Society (SLS)
Former resident at the Miami VAMC and Bascom Palmer Eye Institute 2012-13
University of Montreal School of Optometry graduate ’12
